
Human-centered AI for cancer care
Our AI test helps identify when a repeat prostate biopsy may be needed, supporting physicians in finding aggressive cancers early and helping patients make confident, informed decisions.


Our Mission
At Amorphic, we place the patient at the center of our science. Our mission is to use artificial intelligence to help patients and physicians see the full picture before making the next step.
How It Works
Our AI model analyzes subtle microscopic patterns in negative prostate biopsy slides to predict patient outcomes. The result is a risk score that helps your doctor decide whether a repeat biopsy is necessary - or if you can safely monitor.
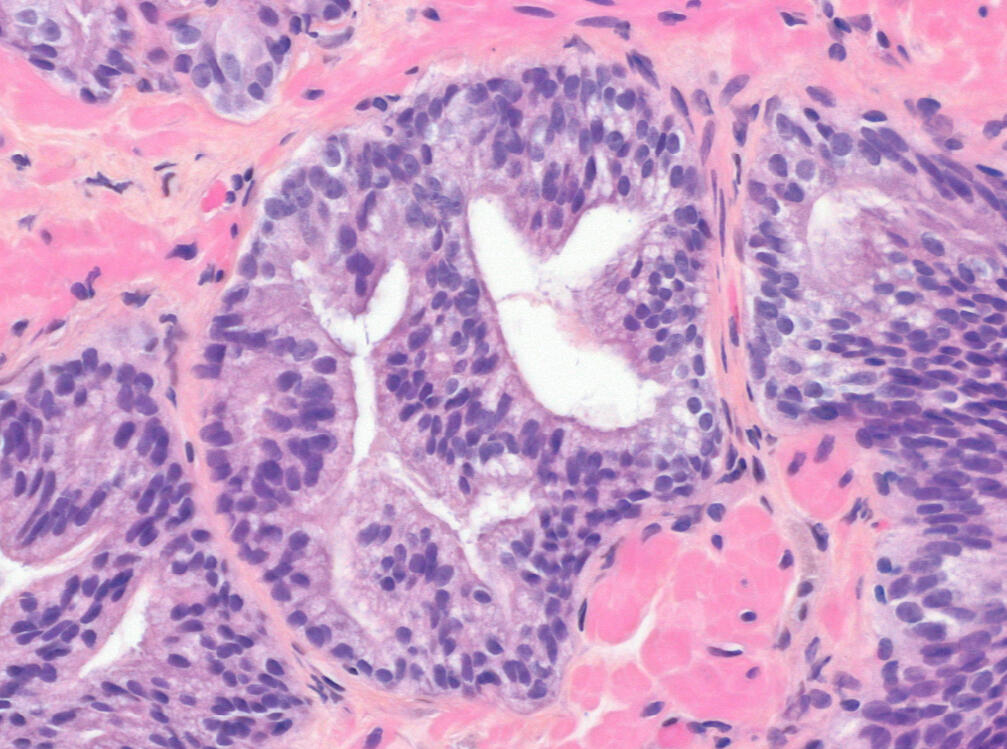
Our Science
Trained on thousands of patient samples, our proprietary prostate foundation models identify subtle, biologically meaningful patterns in tissue that enhance diagnostic precision and clinical confidence.
3.000.000
prostate biopsies performed worldwide in one year. 1
Up to 70%
of prostate biopsies are negative. 2
Up to 34%
reported false negative rate. 3
1. Suzman, Evan; Strasser, Mary Oakley; Kaneria, Aayush; Feinberg, Jonathan; Hu, Jim C.1. Systematic Review and Meta-Analysis on the Timing and Severity of Prostate Biopsy Infections. JU Open Plus 3(11):e00131, November 2025. | DOI: 10.1097/JU9.0000000000000367
2. Rivas, Juan & Alvarez-Maestro, Mario & Czarniecki, Marcin & Czarniecki, S. & Socarras, Moises & Loeb, Stacy. (2017). Negative Biopsies with Rising Prostate-Specific Antigen. What To Do?. European Medical Journal Urology. 5. 76-82. 10.33590/emjurol/10314704.
3. Kawa SM, Benzon Larsen S, Helgstrand JT, et al What is the risk of prostate cancer mortality following negative systematic TRUS-guided biopsies? A systematic review BMJ Open 2020;10:e040965. doi: 10.1136/bmjopen-2020-040965
Find out more
Whether you’re a clinician or a researcher, we’re happy to walk you through how our AI test works.
The Amorphic Prostate AI test is for research use only (RUO) and is not available for diagnostic use.
© Amorphic Lab. All rights reserved.
Thank you
We will contact you shortly with details about our tools and how they may support your clinical research.